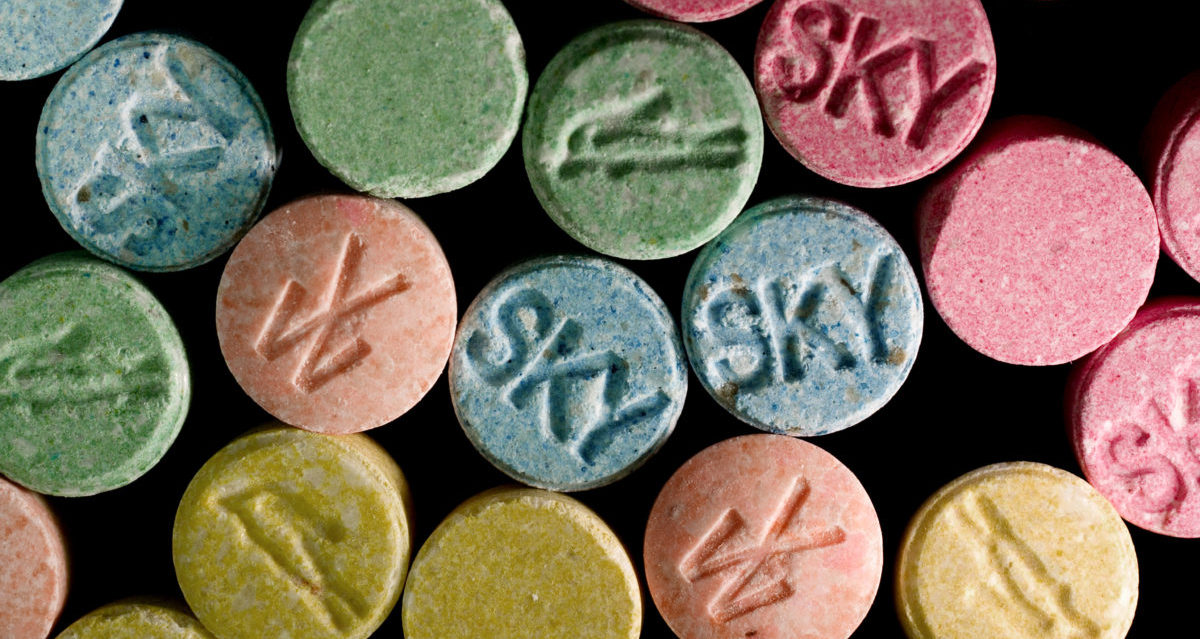
Everyone’s favourite love drug (and the new big thing of recent times) MDMA has been approved by US body the Food and Drug Administration for phase-three clinical trials.
Phase three is the final stage of research trials required before a drug can be considered for a legal prescription drug status.
MORE: Could MDMA be used in couple’s therapy? This academic thinks so
The potential for molly to be used to treat post traumatic stress disorder has been relatively well documented over the years, including a study that dates back to 2011.
Last year the American DEA approved a clinical trial of MDMA to treat anxiety in seriously ill patients.
Earlier this year a leading researcher, Southbank Uni’s Katie Anderson, suggested that molly could be particularly useful when it comes to couple’s therapy.
“In ten years’ time, no one will bat an eyelid if you say you are going to a MDMA-assisted psychotherapy session,” she said.
“I would like to see this stigma fade so that we can conduct the research needed to give us a real, evidence-based picture about the true potential value and harm of illicit drugs.”